Pharmaceutical Distribution Business
We are contributing to reduction in social costs by realizing a stable supply of pharmaceuticals with high distribution quality as social infrastructure and optimizing distribution inventory through the construction of a healthcare distribution platform.
Opportunities and risks
Opportunities
- Expansion of the specialty drug market
- Expansion of generic drug usage
- Spread of digital technologies in the medical and nursing care field
- Increase in digital health services
- Promotion of community-based healthcare collaboration and comprehensive community care systems
Risks
- Stagnant market growth due to controls over increases in national healthcare spending
- Changes in pharmaceutical distribution and sales activities (in response to / in compliance with the guidelines※)
- Increased complexity of distribution inventory management (uneven inventories and pharmaceutical waste)
- The 2024 problem in logistics
- Growing competition due to market entrants from other industries • Circulation of counterfeit pharmaceuticals
- Natural disasters and pandemics
-
※Guidelines for the Improvement of Commercial Transaction Practices:Guidelines for those involved in pharmaceuticals distribution, created by the Ministry of Health, Labour and Welfare in January 2018 to accelerate distribution reform initiatives addressing key issues in pharmaceutical distribution, such as improving unfinalized pricing and provisional supply practices, promoting item-by-item pricing contracts, and eliminating negative primary sales margins. (Revised November 2021 and March 2024)
Suzuken Group strengths
Safe, secure, and reliable pharmaceutical distribution system
- Nationwide sales and logistics networks with a distribution quality that meets global standards
- Nationwide BCP network anchored in Tokyo, Nagoya, and Osaka
- Unification of sales and distribution systems for all Group-affiliated wholesale companies
- Development and placement of GDP specialists
Effective and efficient sales and distribution systems
- Expansion of back-office functions for sales (approximately 50 locations with sales support staff)
- Automation and labor-saving through the use of robot technology and AI
- Cubixx solution, a new digital-based distribution mode
Solid customer relationships
- Network with medical institutions, pharmacies, and medical professionals
- Development and deployment of digital transformation talent
- Stable share of the ethical drug market (based on drug prices):21.5%
Overview of sales and distribution sites and commercial vehicle fleet in the pharmaceutical distribution business (As of March 31, 2025)
- Sales offices:201
- Wholesale distribution centers:15
- Commercial vehicles:Approx. 2,500
- ISO 9001:2015 certified locations (As of January 31, 2026):S.D. Logi Logistics Promotion Department, Sapporo DC, Miyagi DC, Osaka Office, Greater Tokyo DC,Meinan DC, Hanshin DC, Chiba DC, Kanagawa DC,Kakegawa Product Center, Konan DC, SILC (Shoyaku), Okayama DC (Sanki), Niihama DC (Astis), Suzuken Okinawa Yakuhin Headquarters
Main initiatives
Pharmaceutical Distribution Business
Safe, secure, reliable pharmaceutical distribution system
The Suzuken group purchases ethical drugs, diagnostic agents, medical equipment, medical materials, and medical food products from approximately 1,000 companies both in Japan and abroad; including pharmaceutical and medical equipment manufacturers, and supplies medical institutions and insurance pharmacies.
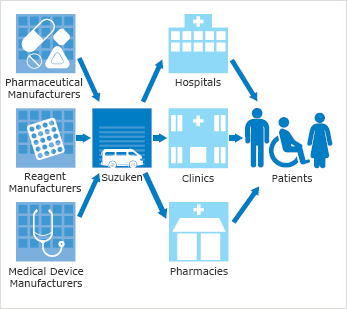
Reliable supply of the required quantity of drug at the required time
The Suzuken group has built a reliable network under strict quality control to deliver the required quantity of drug at the required time to every corner of the country. A nationwide traceability system centrally manages lot numbers and expiry dates and clarifies distribution channels for all pharmaceuticals. This ensures a quick response in the event of a drug recall.
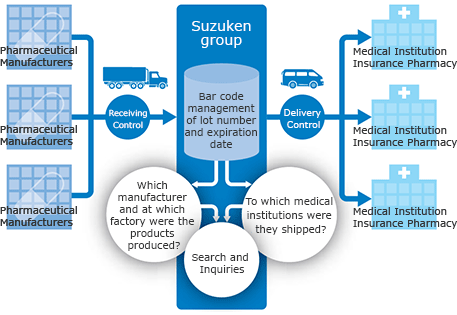
Stable supply system which also serves in emergencies
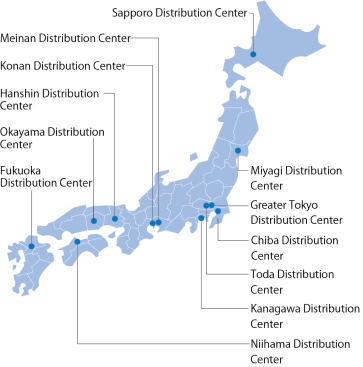
Beginning with distribution centers in 12 locations nationwide and 270 physical distribution bases, the Suzuken group has established a distribution system with an extensive inventory, ensuring that stock does not run out. In preparation for emergency situations such as earthquakes or other large-scale disasters or outbreaks of new strains of influenza, we work on maintaining a stable supply system on a daily basis.


Evolution of healthcare distribution platform
Aiming to become Japan's No.1 medical logistics partner—The Suzuken Group's "Only One" medical distribution platform
The Suzuken group has strengthened its two functions of "Manufacturer distribution" and "Wholesaler distribution". These cover distribution from pharmaceutical manufacturers to pharmaceutical wholesalers and distribution from pharmaceutical wholesalers to medical institutions and insurance pharmacies respectively. We have also built a medical distribution platform with high-quality wide ranging functions unrivaled in the industry, including distribution of investigational and orphan drugs requiring strict temperature control.
In 2012, we became the first pharmaceutical wholesaler to offer comprehensive support services in the field of rare diseases. Using our healthcare distribution platform, we became able to flexibly respond to needs that could not be served by existing distribution systems and made the reliable supply of orphan drugs a reality.
In terms of quality, we comply with the Good Distribution Practice (GDP) guidelines for pharmaceuticals and are strengthening our GDP initiatives through a combination of people and systems, such as obtaining ISO 9001:2015 certification at our wholesale distribution centers and developing in-house certified “GDP Specialists.”
The Group has obtained ISO 9001:2015 certification at 15 locations. In manufacturer logistics, this includes eight logistics centers operated by S.D.Logi(Koga, Sugito, East Japan, Tsukuba, Kobe, Amagasaki, West Japan, and Rokko), together with its Reliability Assurance Office, and Manufacturer Distribution Department, and two sites operated by Chuounyu (Iwatsuki Branch and Headquarters). In wholesale distribution, it covers three sites: Suzuken’s Miyagi Reason for selection (comments from the secretariat): S.D.Logi successfully extended the ISO 9001-based quality assurance system originally implemented in manufacturerlogistics, to a broader wholesale domain, promoting the development of an integrated, companywide healthcare distribution platform. By developing and standardizing key documents such as product management manuals, document control procedures, training programs, change management protocols, and temperature validation guidelines, and by taking the lead in obtaining certification at multiple wholesale sites, the company established a highly reproducible framework. These efforts were highly commended for delivering significant improvements in key quality indicators within wholesale distribution,such as temperature control compliance and delivery accuracy. Going forward, further enhancement of effect analysis is expected to clarify the causal relationship between initiatives and outcomes, paving the way for the evolution of these activities intoTotal Quality Management (TQM). Distribution Center, Osaka Office, and S.D.Logi’s Logistics Promotion Department. We have established an advanced quality management system in compliance with international PIC/S GDP standards.
In addition to the above, we are currently working to obtain ISO 9001:2015 certification for an additional 12 wholesale distribution sites. Going forward, the Group will continue to strengthen its quality management system with the full commitment of the entire organization and continue to fulfill its social mission in pharmaceutical distribution.
-
※1PIC/S: A combination of abbreviations for the Pharmaceutical Inspection Convention (PIC) and the Pharmaceutical Inspection Co-operation Scheme (PICS), which are two international cooperation organizations that aim to improve cooperation among governments and inspection authorities in the areas of GMP and GDP
-
※2GDP (Good Distribution Practice): Quality management guidelines for the transportation and storage of pharmaceuticals
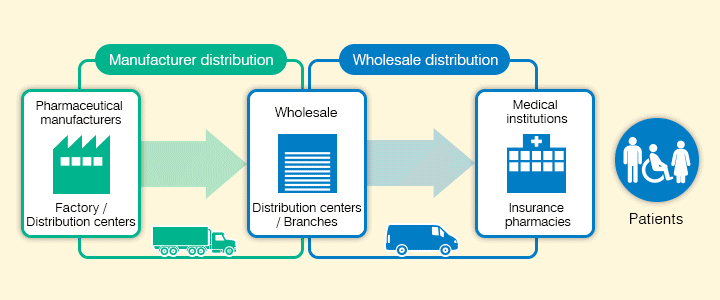

New distribution model for revised product categories
Specialty drug distribution
Since 2017, we have been deploying Cubixx, a specialty drug traceability system. This system uses RFID, which reads and writes data through IC tags, and dedicated storage cabinets equipped with IoT technology to enable 24/7 real-time remote monitoring of pharmaceutical management conditions, from the drugs’ transit to hospital storage, as well as the reverse distribution of returned items and their re-delivery. We have expanded our lineup thus far to include a hospital version, pharmacy version, clinical-trial version, home-use version, large-size version, and room-temperature version.
Monitoring pharmaceutical transit and storage conditions makes it possible to make resale decisions and ascertain inventory conditions, and it contributes to reducing pharmaceutical waste and preventing maldistribution of Construction of healthcare distribution platform utilizing digital means Social issues Reducing social costs by building a healthcare distribution platform utilizing digital means 33 Preparation for New Growth Businesses Strategy Implementation New mechanism enabled by inventory visualization Delivery schedule app, delivery schedule notification service Ordering support Automatic route creation Forecasts future demand based on delivery records, and alerts to orders and returns Allows online checking of the delivery date for ordered products, as well as the availability of substitutes Creates optimal delivery routes for each day, including returns and collections inventory. It also enables automated ordering and allows automation of things like proposals to refresh non-moving inventory or unused inventory, which helps to reduce the workload on medical institutions.
Responding to needs for community-based healthcare collaboration and comprehensive community care systems
The Suzuken Group is working to connect people and resolve issues in local healthcare and nursing care through partnerships with pharmacies and nursing care businesses, and collaborations with IT-driven healthcare companies. Already, we have entered into partnership agreements with numerous local governments at the branch, sales department, and company level in an effort to move forward with contributions to local communities.

Healthcare Distribution Reform
Initiatives to improve distribution and productivity
We are promoting healthcare distribution reform with the aim of securing the appropriate profits necessary to maintain the functioning of social infrastructure. In this context, we are adhering to the Guidelines for the Improvement of Commercial Transaction Practices and working on cost structure reforms, including promoting a shift in employee mindset toward profit-oriented management. In addition, as part of establishing a sales system that combines real-world and digital elements, we are promoting the use of COLLABO Portal, the Delivery Schedule Notification Service and Delivery Schedule Notification app, and the Order Proposal app. We are also expanding back-office functions, in order to maximize the real-world customer touchpoints for which MS are responsible and to improve productivity.
Combining real and digital means to increase customer contact points
The conventional pharmaceutical wholesale system relied on sales representatives known as MS, constantly requiring their judgment and confirmation for everything from orders to deliveries and returns. Considering that MS will be more important with the transition to a health creation enterprise, we will conduct healthcare distribution reform and build an effective and efficient sales mechanism to transition from pharmaceutical wholesaling to healthcare distribution.
Specifically, we will strengthen three backyard functions: the “Sales support functions,” by building a support mechanism that takes over the indirect operations of MS and also by utilizing the services of COLLABO Portal; “Customer functions,” by utilizing an app that visualizes inventory data and delivery schedules, and by building a customer center; and “Logistics functions,” by partnering with our Group company S.D. Logi CO., LTD. This will help create an environment where MS can concentrate on their original roles of connecting the Group and its customers, widening the scope of health creation, and creating revenue.
When it comes to sales activities, we will continue to value the conventional way of building relationships of trust with customers through visit-based, in-person sales, while also expanding digital points of contact, such as by providing information and proposing solutions through COLLABO Portal. While optimizing the number of visits, we will also emphasize efficiency and maintain the amount of contact points with customers.
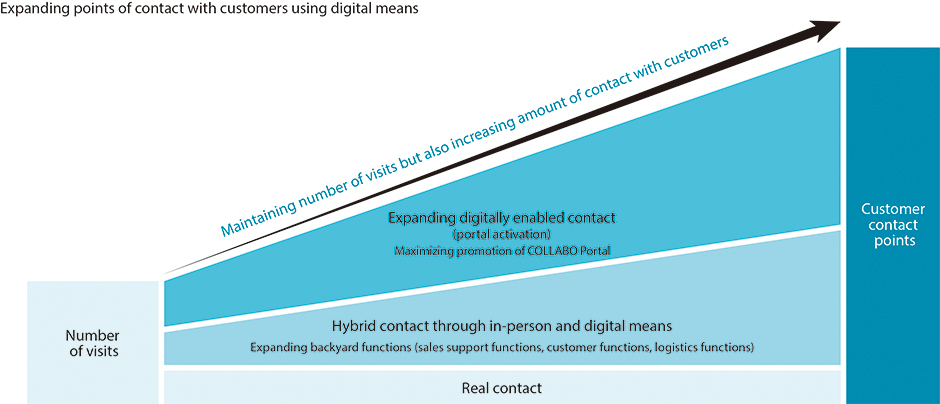
Constructing new sales mechanisms
By introducing digital and backyard functions into our sales activities, we will construct two sales mechanisms: “hold-fast sales” via the healthcare distribution platform that acts as social infrastructure, and “new sales” that provide new value. For “hold-fast sales,” we will leverage the relationships of trust cultivated thus far with customers, along with the Suzuken Group’s ability to mobilize, to help with problem solving on the front lines for customers and communities.
At the same, we will promote COLLABO Portal to fulfill the coordinator role of connecting the Group with customers and communities. In “new sales,” we will propose solutions to key personnel in medical and nursing care and the community utilizing digital health services posted on COLLABO Portal.
In the future, we aim to provide new value and contribute to not only medical and nursing care professionals, but local society as well.
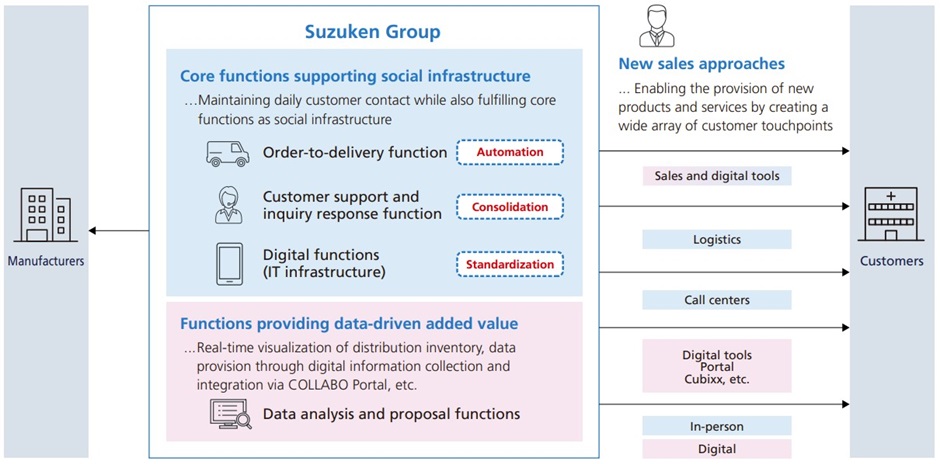
Main solutions resulting from collaborations
- COLLABO Portal, a comprehensive platform for medical digital transformation
- COLLABO Mobile, a medical and nursing care device terminal provision service
- The Cubixx specialty drug traceability system
- PS+Management inventory management system for pharmacies
- PS+Voice automatic voice recognition and electronic medication history system
- Medifiims PE asset management system for pharmacies
- ES Navigation, a remote detail service
- Digital services: Medical Care Station (MCS), a communication tool dedicated to medical and nursing care professionals Dr. JOY medical institution support platform, Welby MyKarte PHR (Personal Health Record) platform, Ubie for Hospitals AI-based medical interview service, SmartMat, an IoT order and inventory management solution
- ARUU, a prescription drug delivery service
Asian business
Rollout of pharmaceutical distribution business in China and South Korea
Constructing medical platform in China
In Asia, national governments are moving ahead with healthcare system reforms in response to their growing populations and rising economic performance. The implementation of advanced, efficient pharmaceutical distribution is a high priority and there are great expectations for how the Suzuken Group, and the experience and knowledge it has developed in Japan, can contribute.
China, a country of particular interest, with developments such as rising incomes and the institution of a healthcare insurance system, is now the world’s second largest pharmaceutical market.
Suzuken has been developing pharmaceutical distribution operations in Shanghai and Qingdao by establishing in 2008, a joint venture (49.9% ownership), Shanghai Suzuken Huzhong Pharmaceutical Co., Ltd. (now SPH Suzuken Huzhong (Shanghai) Pharmaceutical Co., Ltd.), with Shanghai Pharmaceutical Co., Ltd., a major Chinese pharmaceutical wholesaler.
Furthermore, in September 2016, Suzuken acquired a 35% stake in EPS EKISHIN Co., Ltd.,※6 a member of the EPS Group. Together, the two are helping Suzuken (Shenzhen) Pharmaceutical Co., Ltd. (20% stake held by Suzuken) to introduce the products of Japanese pharmaceutical companies to the Chinese market and are performing technical sales promotion activities throughout China on behalf of Suzuken (Shenzhen) Pharmaceutical.
Working with its partners, Suzuken draws on R&D, clinical trial, manufacturing, sales, sales promotion, distribution, and collection functions to create a healthcare distribution platform capable of providing customers with the one-stop services they need. Looking to the future, Suzuken will continue to create new added value for the field of healthcare in China.

-
※6EPS EKISHIN Co., Ltd. is a specialized trading company connecting Japan and China in the field of healthcare. Its wide-ranging business pursuits span areas as varied as CRO services, pharmaceutical manufacturing, and sales agency for digital diagnostic imaging equipment.
Constructing national distribution network for healthcare products in South Korea
In South Korea, as in other countries, healthcare expenditures are on the rise and there are expectations that the healthcare industry will continue to advance and that the government will lead policy efforts to curb healthcare expenditures.
Suzuken entered into a capital and business alliance with BOKSAN NICE Co., Ltd.※7 taking a 45% stake in that company in June 2016. Drawing on knowledge it has developed in the field of health creation in Japan, the Suzuken Group is supporting BOKSAN NICE in its efforts to expand its pharmaceutical distribution business to cover all of South Korea and advance joint R&D in medical-care related businesses. The Suzuken Group is also helping BOKSAN NICE to enhance its corporate value through joint projects for strengthening its distribution functions and management platform.
By continuing to partner with local companies, we are also contributing to the advancement of pharmaceutical distribution and industries related to medical care in South Korea.

-
※7BOKSAN NICE Co., Ltd. with operations mainly in Busan Metropolitan City and the Seoul Capital Area, is a pharmaceutical distribution leader in South Korea.
Building a new network for further development of the South Korean pharmaceutical industry
In May 2025, we entered into a business alliance with DONGWON Group, which operates a pharmaceutical distribution business in South Korea, and a capital and business alliance with KYONGNAM DONGWON Pharmaceutical Wholesale Co., Ltd., a subsidiary of the same group. Through collaboration between BOKSANNICE and DONGWON Group, which stems from our alliances, the two companies will share their procurement and logistics functions, expand into the untapped southwestern region of South Korea, and build a network that delivers healthcare products across the country.
By advancing initiatives together with local partners who share our vision to contribute to resolving social issues, the Group will continue contributing to the growth and development of South Korea’s pharmaceutical-related industry.

Expanding existing services and discovering new services with Cencora
With the aim of expanding business in Asia, primarily in China and South Korea, we are deepening our partnership with major US wholesaler Cencora (former AmerisourceBergen) and considering the possibility of rolling out the Cubixx system. Also, through a partnership between our Corporate Venture Capital (CVC), which we established in 2022 to facilitate the Suzuken Group’s investment in health-tech companies, and Cencora’s CVC, AB Health Ventures ($150 million), we will discover new technologies and solutions in the global healthcare field and consider their introduction in Japan and Asia.
Related group companies and affiliates
Subsidiaries
Pharmaceutical Distribution Business
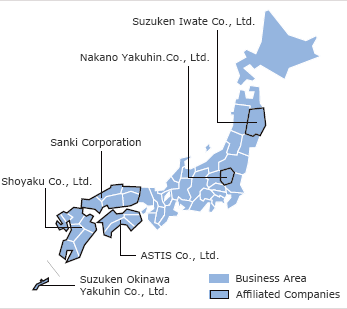
- Suzuken Co., Ltd.
- Sanki Corporation
- ASTIS Co., Ltd.
- Shoyaku Co., Ltd.
- Suzuken Okinawa Yakuhin Co., Ltd.
- Nakano Yakuhin Co., Ltd.
- Suzuken Iwate Co., Ltd.
- S.D.Logi CO., LTD.
- Cloumd Corporation
- PSC Co., Ltd.
- Sanki MediHeart Limited
Associated Companies
Asian Business
- SPH Suzuken Huzhong (Shanghai) Pharmaceutical Co., Ltd
- BOKSAN NICE Co., Ltd.
