Healthcare-Related Services Business
We provide manufacturer support services such as contract distribution for manufacturers and contract distribution of orphan drugs to meet diverse needs of pharmaceutical manufacturers in Japan and overseas, and digital healthcare services to solve issues in the healthcare field.
Opportunities and risks
Opportunities
- Growing demand for high quality cold chain logistics
- Growing 3PL market from the diversification of logistics needs
- Growing demand for outsourcing
- Creation of IT platforms in logistics (streamlining)
- Increase in digital health services
- Integration and utilization of healthcare data
Risks
- Mandatory compliance with GDP standards in pharmaceutical distribution
- Rising cost associated with GDP compliance
- The 2024 problem in logistics
- Growing competition due to market entrants from other industries
- Revision of Guidelines for the Improvement of Commercial Transaction Practices
- Rising costs of preparing for disasters and pandemics
- Shortage of logistics personnel and medical and nursing care professionals
- Growing demand for reducing environmental impact across the supply chain
Suzuken Group strengths
Know-how and track record in manufacturer distribution and specialty drug distribution
- Supply chain based on comprehensive Group strengths
- Greater shipping efficiency and optimization of distribution inventory through shared distribution
- GDP-compliant quality management and nationwide shipping and delivery network
- Development and deployment of GDP specialists
- Expertise and track record in cold chain logistics
- Manufacturer distribution service (as of March 31, 2025): 49 client companies
Locations of the manufacturer distribution service (As of March 31, 2025)
- Manufacturer distribution centers:11
- ISO 9001:2015 certified locations (as of January 31, 2026): S.D. Logi Manufacturer Distribution Department, Kobe Distribution Center (DC), Koga DC, Amagasaki DC, Sugito DC, West Japan DC, East Japan DC, Rokko DC, Tsukuba DC, Sapporo DC
Customer support through digital technologies
- Registrations on the COLLABO Portal, a comprehensive platform for medical digital transformation (As of March 31, 2025):Approx. 300,000 user accounts
Main initiatives
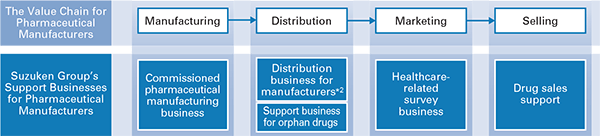
Manufacturer Support Services Business
Providing a variety of services to pharmaceutical manufacturers
We contribute to pharmaceutical manufacturers' cost reductions by contracting pharmaceutical manufacturing, operating manufacturer distribution centers and contracting wholesaler distribution. Further, we regard it as our social mission to contribute to the distribution of orphan drugs※1, ensuring that necessary drugs are delivered even if there is only one patient who suffers from an illness.
-
※1Drugs with high medical needs but required by only a small number of patients
-
※2Distribution for manufacturers: In the above diagram, "procurement distribution" means transportation of pharmaceutical raw materials to the manufacturer's factory; "production distribution" means transportation of pharmaceuticals from the manufacturer's factory to its warehouses; and "sales distribution" means transportation of pharmaceuticals from the manufacturer's warehouses to our wholesale distribution centers.
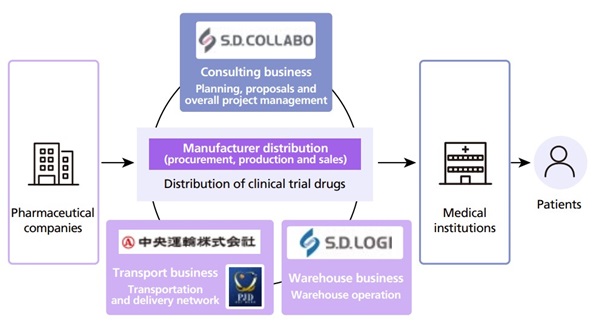
Providing high-quality logistics services through Group-wide capabilities
In 2005, we became the first pharmaceutical wholesaler in Japan to provide contract distribution service for manufacturers. Today, we operate 11 manufacturer distribution centers, handling pharmaceuticals and related products equivalent to 2.6 trillion yen annually (based on drug prices). By establishing an integrated system that manages both manufacturer and wholesale distribution, we have consolidated logistics services that were previously managed separately by individual pharmaceutical companies, thereby improving delivery efficiency, optimizing distribution inventory, and contributing to the reduction of social costs.
We leverage the comprehensive strengths of the Suzuken Group. These include consulting support for pharmaceutical companies provided by S.D. COLLABO Co., Ltd., warehouse operations by S.D.Logi CO., LTD., and a nationwide transportation and delivery network led by CHUOUNYU CO., LTD. Through this integrated approach, we provide high-quality services that meet needs throughout the entire supply chain and have established ourselves as the logistics partner of choice for pharmaceutical companies.
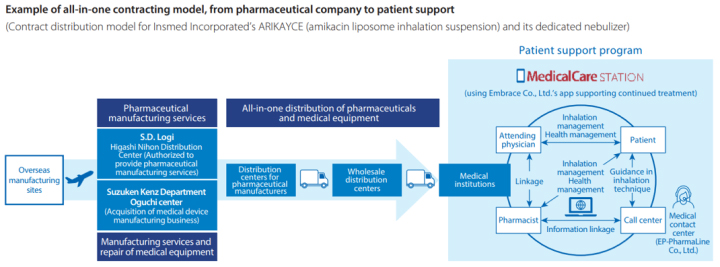
Functional expansion of our all-in-one contracting model
In recent years, the number of foreign pharmaceutical companies aiming to enter the Japanese market has been increasing, but many have no manufacturing plants, distribution centers, or a distribution network. To meet these needs, in April 2021, we entered into a collaboration with Bushu Pharmaceuticals Ltd., and combining the capabilities of our two companies has allowed us to undertake the all-in-one contract provision of specialty drug services ranging from marketing authorization holders’ consulting to import, inspection, manufacturing, distribution, post-marketing surveillance, and patient support. Additionally, we have a manufacturing operations function at a distribution center owned by Chuounyu Co., Ltd., a member of the Suzuken Group. We have absorbed EP-PharmaLine Co., Ltd., a subsidiary of EPS Holdings, Inc., with whom we began a capital and business alliance in 2016. And we integrated EP-PharmaLine’s medical contact center functions and BPO functions, for example, with the Suzuken Group’s functions to build a platform that meets pharmaceutical companies’ needs all in one stop.
In January 2023, in partnership with pharmaceutical companies, we started the industry’s first joint shipping, in compliance with GDP guidelines. By improving the efficiency of distribution from pharmaceutical companies to wholesale distribution centers, we are also helping to reduce the environmental burden.
Realizing Quality Management in Line with Global PIC/S GDP Standards
In our contract distribution business for pharmaceutical manufacturers, which the Suzuken Group began providing in 2005, we practice strict temperature and quality control in conformance with GMP※3 standards. At our manufacturer distribution centers, we gained GMP-compliant ISO 9001 certifications in 2008, have updated those certifications to the 2015 version, and provide quality control in conformance with the PIC/S※4 GDP※5 global standards. We are now expanding the expertise cultivated in our manufacturer distribution operations to our wholesale distribution centers, in an effort to strengthen end-to-end quality management. With the start of operations at CHUOUNYU CO., LTD.’s Medical Terminal (photo on page 42) in October 2018, more efficient joint pharmaceutical distribution is now being provided in the Kanto Area. In addition, efforts are being made to expand the area in which we are able to offer our Direct Cool※6 refrigerated distribution service for pharmaceuticals.


-
※3GMP (Good Manufacturing Practice): Manufacturing and quality management guidelines for pharmaceuticals
-
※4PIC/S: A combination of abbreviations for the Pharmaceutical Inspection Convention (PIC) and the Pharmaceutical Inspection Co-operation Scheme (PICS), which are two international cooperation organizations that aim to improve cooperation among governments and inspection authorities in the areas of GMP and GDP
-
※5GDP (Good Distribution Practice): Quality management guidelines for the transportation and storage of pharmaceuticals
-
※6Direct Cool: Temperature controlled service in which pharmaceuticals (2~8°C) are shipped from Chuounyu Medical Terminals throughout the country directly to pharmaceutical wholesale distribution centers
S.D.Logi Wins TQM Growth through Challenge Award for FY2025
Strengthening of distribution platform for CGT products
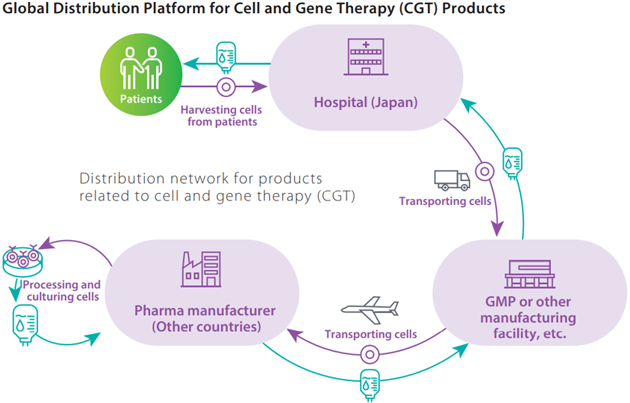
In 2016, we initiated the distribution of clinical trial products for cell and gene therapy (CGT), and we collaborated in 2018 with World Courier, a part of the U.S.-based Cencora (former AmerisourceBergen) drug wholesale group, to create a global network for CGT products. We are expanding our contract distribution services for pharmaceutical companies that aim to enter the Japanese market and launch new products, such as Novartis Pharma K.K.’s Kymriah, Japan’s first CAR-T cells medication, and ZOLGENSMA IV infusion, a gene therapy product for treating spinal muscular atrophy, Together with SanBio, Inc., we jointly developed the R-SAT support system for CGT products distribution management and drug administration scheduling, and we obtained a patent for it in April 2022. In addition, through initiatives such as obtaining manufacturing authorization for storage, labelling, and decision-making for product shipping, a first for a pharmaceutical wholesaler, we are expanding our services for responding to the sophisticated, diversifying needs of pharmaceutical companies.
Digital Services Business
Creation of digital services through collaboration with health-tech companies
Since 2019, the Suzuken Group has been actively advancing partnerships with health-tech companies. After entry into capital and business alliances with Dr. JOY Co., Ltd., which offers efficiency solutions for healthcare settings, and with Welby Inc., a leading company in personal health record (PHR) services, we formed a similar alliance with Ubie, Inc. in April 2020 and embarked on the joint promotion of the Ubie for Hospitals medical interview service for medical institutions. In November 2020, we entered into a capital and business alliance with Doctors Inc., a company building and promoting a healthcare DX platform from the perspective of frontline healthcare professionals. April 2021 saw the addition of Embrace Co., Ltd., a capital and business partner since February 2020, as a Suzuken subsidiary, and now we are developing business based on a platform applying Medical Care Station (MCS). In November 2021, we entered into a capital and business alliance with FRONTEO, Inc., which operates a data analysis business using artificial intelligence (AI). We are conducting distribution-related activities on an exclusive basis for the conversational dementia diagnosis support AI program that FRONTEO provides, and we aim to establish a system to quickly penetrate and expand the market.
In February 2022, we began building a new added value service for the Cubixx System with SUSMED Inc., with whom we formed a capital and business alliance in May 2020. We will leverage that company’s digital healthcare platform, which consists of blockchain technology for preventing data falsification and identity theft, its AI-based automated analysis systems, and other technologies, to enable communication activities and the optimization of market inventory management.
Together with a variety of companies, we will launch digitalization efforts to solve the problems that the industry is facing and to build a safe and secure healthcare platform, thereby contributing to patients and to community healthcare and the healthcare professionals that support it.
Establishment of new companies to promote digital business
With the increase in digital healthcare services, we believe that a new “digital wholesaler” function is necessary to manage the distribution of healthcare apps, programs, and other digital materials. Making the best use of our Group strengths as “connecting capability” and “spreading capability,” we are now developing a comprehensive, all-in-one portal site that seamlessly “connect”s doctors, pharmaceutical companies, and patients with a variety of digital healthcare services from both Japan and overseas which we will “spread” on the platform. We will ensure that this site contributes to customers and community-based healthcare and provides optimal solutions to pharmaceutical companies.
In addition, we are looking into establishing a Health-tech Collaboration Council (tentative name) with the goal of ensuring that a safe, secure, and correct concept of DX penetrates and takes root, one that creates a level of security and compliance that serves as the industry standard. Furthermore, we set up a Corporate Venture Capital (CVC) fund to shift investment in health-tech companies into full swing in April 2022. We will partner with a variety of health-tech companies that have leading-edge technology, business models, and ideas, to create new solutions.
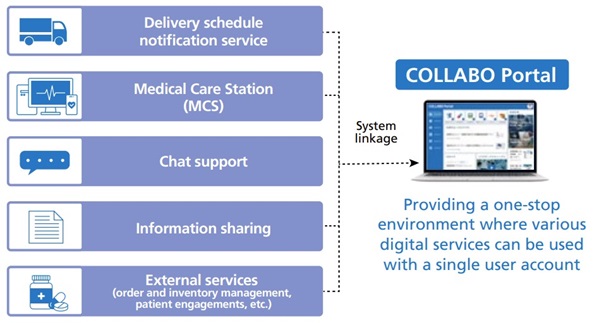
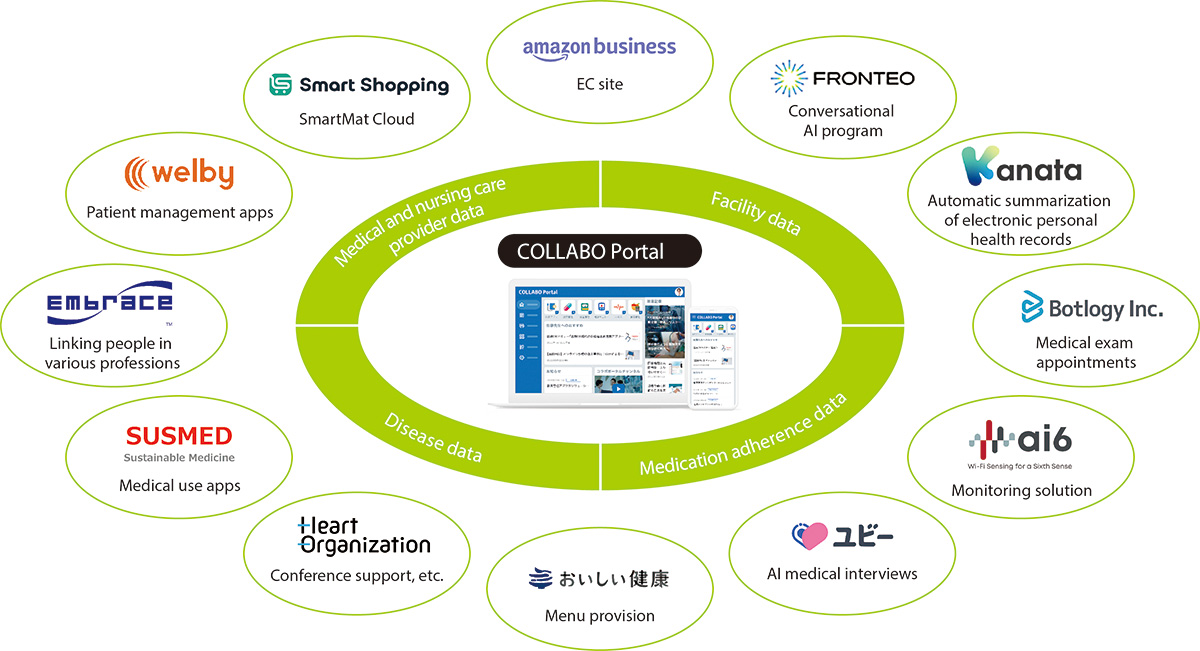
Development of a digital platform based on the COLLABO Portal
In April 2023, we launched the COLLABO Portal, a comprehensive platform for medical digital transformation. The platform serves as a hub connecting manufacturers, medical and nursing care professionals, patients, and their families, and is currently utilized by approximately 300,000 medical and nursing care professionals. The portal enables users to access digital health services from our company and partner companies in one place, and also allows for direct inquiries within the platform. This contributes to improving the quality and efficiency of the work of medical and nursing care professionals.
Going forward, we will continue to promote further expansion of COLLABO Portal, linking and leveraging the accumulated data to support the creation of new products and services by a variety of manufacturers, including pharmaceutical companies. Ultimately, these efforts will contribute to enhancing access to medical and nursing care services and improving the quality of life for patients and their families, who benefit from these services.
We also began rental services in June 2023 for COLLABO Mobile, a communications device that will enable medical and nursing care providers to access digital health services all in one place when needed. COLLABO Portal’s functions are installed as standard on COLLABO Mobile, and it can be used not only in facilities but also at patients’ homes and other home healthcare and nursing care sites in the community.
Popularization and expansion of services in preparation for digital business rollout
Centering on COLLABO Portal, we will roll out a wide range of services that support medical and nursing care providers. Utilizing the services-related information and patients’ ailments and medication data that are accumulated there, we aim to create services that meet diversifying needs and to realize a platform that is appealing to medical and nursing care providers.
Related group companies and affiliates
Subsidiaries
Healthcare-Related Services Business
- CHUOUNYU CO., LTD.
- Net Hospital, Inc.
- P.J.D. Network
- JIT Co.,Ltd.
- S.D. Collabo Co., Ltd.
- Suzuken Business Associe Co., Ltd.
- Embrace Co., Ltd.
- Galenus Co., Ltd.
- Life Support Co.,Ltd.
- Suzuken JoinUs Co., Ltd.
- Collabo Square Co., Ltd.
- Suzuken Investment Co., Ltd.
- Kyuyaku Service Limited
Associated Companies
Healthcare-Related Services Business
- EP-PharmaLine Co., Ltd.
- Welby Inc.