Healthcare Product Development Business
We conduct research and development, manufacturing, and sales of ethical drugs, diagnostic reagents, and medical devices and materials that contribute to improvement of the quality of life of patients and unmet medical needs.
Opportunities and risks
Opportunities
- Change in disease patterns due to aging population and other factors (growth of the market for antidiabetic agents)s
- Response to unmet medical needss
- Growing demand for outsourcing among pharmaceutical companiess
- Increase in diverse startups and new outsourcing service providerss
- Diversifying needs for in-home medical care and nursing cares
- Advancement of digitalization
- Diversification of drug discovery modalities
Risks
- Cuts in drug prices driven by rising medical costs
- Advancement of globalization
- Growing competition due to market entrants from other industries
- Compliance with guidelines for provision of sales information
- Environmental risks in research and development activities
- Drug loss and drug lag in Japan
Suzuken Group strengths
Efficient and high-quality production technology
- Presence in the fields of diabetes, kidney diseases, and dialysis treatment
- Track record of development and manufacturing of pharmaceuticals, diagnostic reagents, and diagnostic devices
- Supply chain management system that ensures a stable supply of highquality products
- Advanced drug production system that is compliant with international GMP standards
- Track record in contract manufacturing for major pharmaceutical companies
- OSDrC (One-Step Dry Coating) business utilizing proprietary formulation technology
- 100% domestic stethoscope manufacturing (domestic market share:approx. 40%)
- Obtained ISO 13485 certification for the quality management system of medical devices
- Extensive track record in the OEM business for medical devices
Comprehensive support system to help pharmaceutical companies place products on the market
- J-ENTRY Consortium, a one-stop service for overseas pharmaceutical manufacturers entering the Japanese market
- Networks with overseas pharmaceutical companies and other entities
Locations of the healthcare product development business (As of March 31, 2025)
- Pharmaceutical manufacturing plant (Kumamoto)
- Pharmaceutical research laboratory (Mie)
- Medical equipment manufacturing plant (Gunma)
Main initiatives
Pharmaceutical Manufacturing Business
Boosting our presence in diabetes therapies and kidney dialysis
We develop, manufacture and sell pharmaceuticals that contribute to the improvement of lifestyle-related diseases with a focus on diabetes, high added value generic drugs, blood glucose self-monitoring devices and diagnostic agents with a top share of the domestic market. Some of our factories conduct contract manufacturing for major pharmaceutical manufacturers. Sanwa Kagaku Kenkyusho's own formulation technology has earned high evaluation at home and abroad.

Manufacturing of ethical drugs
At Sanwa Kagaku Kenkyusho Co., Ltd. we give serious consideration to patient QOL and economic aspects, and develop easy-to-use, safer and more effective medicines.



Manufacturing of diagnostic agents
At Sanwa Kagaku Kenkyusho Co., Ltd. we manufacture essential high-quality diagnostic drugs and diagnostic equipment for ultrasonography, electrocardiography and for testing blood and urine specimens in hospitals and clinics.

Contract manufacturing of pharmaceuticals
At Sanwa Kagaku Kenkyusho's Kumamoto Factory, we carry out contract manufacturing for major pharmaceutical manufacturers in addition to manufacturing our own pharmaceuticals.
Investment to further expand development pipelines
As a pharmaceutical manufacturer, our ongoing growth relies on the development of new pharmaceuticals. We view diabetes, and kidney and dialysis-related areas, as areas with substantial unmet medical needs and strong potential as new growth markets. These areas are at the center of our efforts to develop new pharmaceuticals and enhance our pharmaceutical development pipeline.
In August 2021, we launched the UPASITA IV Injection Syringe for Dialysis, a therapeutic agent for secondary hyperparathyroidism, and we are co-promoting it with Kissei Pharmaceutical Co., Ltd., in an effort to penetrate the market. In February 2022, we concluded a license agreement with Crinetics Pharmaceuticals, Inc.※1 relating to the exclusive development and commercialization of Paltusotine※2 in Japan, and we will move forward on development.
Promotion of patient-centered new product development and introduction
In February 2022, SKK signed a license agreement for exclusive development and commercialization rights in Japan for SK-5307 (Paltusotine), a treatment drug for acromegaly and neuroendocrine tumors. Phase I trials for SK-5307 were completed in June 2023, and a clinical trial notification for Phase II/III trials targeting patients with acromegaly and pituitary gigantism was submitted on October 31, 2023. Furthermore, we signed a strategic collaboration agreement for nucleic acid drug discovery with Nissan Chemical Corporation in April 2024, and also signed an exploratory joint research agreement with Nagoya University in April 2025. Through these and other initiatives, we aim to develop pharmaceuticals that address unmet medical needs.
-
※1Crinetics Pharmaceuticals, Inc.: A clinical-stage pharmaceutical company that focuses on discovering, developing, and commercializing new therapies for people with rare endocrine diseases or endocrine-related tumors
-
※2Paltusotine (tentative name): A nonpeptide, orally bioavailable agonist that has high somatostatin receptor 2 (SST2) selectivity and curbs growth hormone secretion
New business creation through integration of Group functions and collaborations
We are reviewing and restructuring Sanwa Kagaku Kenkyusho’s supply chain as a whole. Taking into account factors such as the direction of new drug development and the increasingly globalized business environment, in September 2022, we transferred the Fukushima Factory, a pharmaceutical manufacturing business location, to Bushu Pharmaceuticals Ltd., which has acquired GMP certification in several countries around the world. We will be jointly employing sophisticated technology and production operations.
Drawing on the strengths we have cultivated in our pharmaceutical manufacturing business—high quality, reliable supply, and low cost—the Suzuken Group aims to create new business models and enhance productivity by combining these strengths with its other capabilities and collaborating with partner companies.
In addition to expanding our flagship diabetes treatment products, we signed a co-promotion agreement with Abbott Japan in January 2024 to enhance care for diabetes patients through our glucose self-monitoring devices.
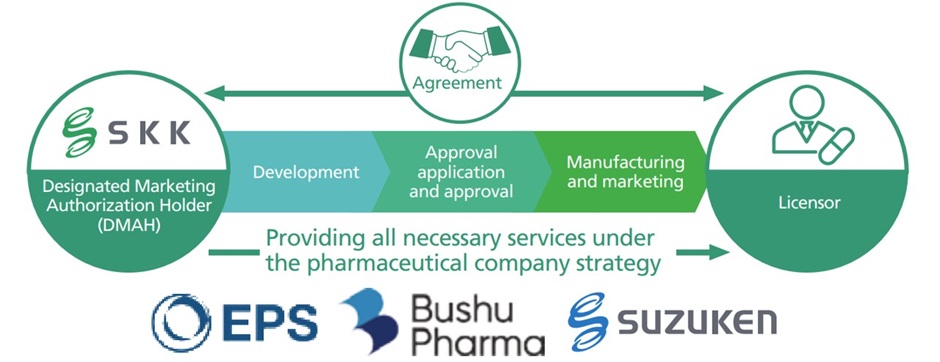
Supporting Japanese market entry for drugs not yet approved in Japan (drug loss items)
In May 2024, we initiated a partnership with Bushu Pharmaceuticals Ltd. and EPS Holdings, Inc., establishing J-ENTRY Consortium, a collaboration model designed to support the entry of unapproved drugs (known as drug loss items) into the Japanese market. We are currently advancing our activities by, for example, introducing this service to overseas biopharmaceutical companies and submitting applications for the development of unapproved and offlabel drugs with high medical needs, in response to public solicitations from the Ministry of Health, Labour and Welfare for drug development companies.
Medical Equipment Manufacturing Business
Pursuing the development of products that are "patient-friendly", expanding globally
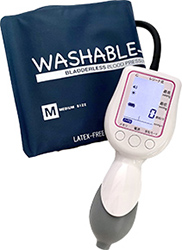
Our core medical equipment whose brand name is "Kenz" developed by Suzuken group is testing equipment such as Holter electrocardiographs, blood pressure monitors and stethoscopes. We are boldly taking on challenge to develop the equipment to make testing easier and more accurate as well as no burden and anxiety to patients and have realized the miniaturization of Holter electrocardiographs.
We have constructed systems including the "ISO13485" quality assurance system for medical equipment to meet global quality requirements. Kenz products are used in medical practice in over 30 countries worldwide, mainly in the Asian region, and have received high evaluation from medical institutions around the world.

Expansion of medical equipment and supplies manufacturing field, and strengthening of our competitiveness in it
Kenzmedico Co., Ltd. leverages the Group’s sales marketing capabilities to develop and manufacture not only the Kenz-brand medical devices and supplies sold through Suzuken but also medical devices such as stethoscopes and sphygmomanometers for professional use. It is the only company in Japan that manufactures stethoscopes entirely domestically, capturing around 40% of the domestic market. In February 2022, we obtained medical equipment certification and insurance coverage for the “Simple Holter,” a test kit that integrates a single-use waterproof Holter electrocardiograph and an electrocardiogram data analysis service. This service is easy to use even at medical institutions that do not have testing experience.


Developing new services and products that leverage points of contact with patients
Pharmaceutical manufacturer Sanwa Kagaku Kenkyusho Co., Ltd. and medical equipment company Kenzmedico Co., Ltd. are in charge of the Suzuken Group’s healthcare product development field. Moving forward, in addition to the product development being carried out in each company’s respective fields, we will combine the functions of our partner companies to provide support for getting specialty products of companies newly entering Japan onto the Japanese market, as well as to propel the development of new products and services such as highly unique pharmaceuticals and medical equipment that leverage synergies among the Group. We will also start developing products and services in additional fields such as early diagnosis, nursing care, and terminal stage medical care.
A strength of the Suzuken Group is that our pharmacy and nursing care business operations serve as direct points of contact with patients, their families, and medical and nursing care providers, which allows us to collect those people’s opinions. Based on the needs ascertained from our customers through the relationships we have cultivated thus far with customers and partner companies, we will link with partner companies from the planning and design stage to contribute to things like market research on products and services.
